Day 2 :
Keynote Forum
Dave Anderson
Syte Logix, Inc., USA
Keynote: Integrating patient generated and 3rd party data into clinical research
Time : 10:00-10:30

Biography:
Dave Anderson is the Founder, CEO and President of Syte Logix. As a healthcare analytics and security executive, he brings his extensive experience in data analytics, cyber-security, risk management, patient privacy, and connectivity to the growing challenges of the MedTech and clinical data management market. His analytic and data exploration models create new opportunities for customers and partners to significantly improve the ROI around clinical research, and get products to market in a faster and more secure manner.
Abstract:
The increasing volume of patient generated and 3rd party data can provide great value throughout the clinical workflow. However, as this data is increasingly unstructured, and is generated from very disparate sources in a variety of formats, it is very difficult to cost-effectively extract the value from this data into the research process. The Opportunity: The integration of patient generated and 3rd party data into the clinical research workflow can add a multitude of new dimensions into all phases of a research program, including drug discovery and development, patient recruitment, risk-based monitoring, and pharmacovigilance. Automating the ingestion, management and exploration of these data sources can help analysts gain deeper context to make better and more cost-effective clinical decisions and uncover new insights that can drive competitive advantage. In this session, we will navigate strategies to integrate and capture the value of patient generated and 3rd party data into the clinical workflow
Keynote Forum
Khalid Abou Farha
Clinical Pharmacology Research Centre, UK
Keynote: Clinical trials in cystic fibrosis patients; does tachycardia matter?: A case report
Time : 10:30-11:00

Biography:
Khalid Abou Farha is medical director and clinical pharmacologist of Celeron he brings 15 years of clinical research experience to this role, spanning a wide range of therapeutic areas, with a particular focus in oncology, vaccines, cancer immunotherapy, personalized medicine and autoimmune disorders. He has extensive experience in building and managing global teams, leading the development of Clinical Research drugs and biologics
Abstract:
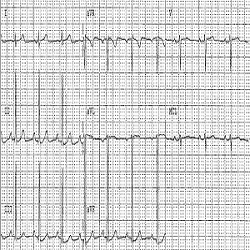
Background: Clinical trials in cystic fibrosis (CF) patients might include post-B2 agonist bronchodilator (e.g. Salbutamol) evaluation of pulmonary functions. Sinus and SVT tachycardia have known dose-related side effects of inhaled salbutamol. A relationship between inhaled beta 2 agonists and increased risk of myocardial ischemia (MI) and infarction has been reported. Under normal breathing conditions, CF patients might show mild tachycardia compared to healthy subjects. Nevertheless, the impact of B2 agonists on the CF- associated tachycardia is often an ignored safety parameter in CF clinical trials. Here, we demonstrate a case of post-salbutamol clinically significant tachycardia associated with ECG features of MI in a CF patient.
Case Report: A 21 year old CF female patient has been included in a clinical trial evaluating a new medication for the treatment of CF. Medical history, physical examination, vital signs and laboratory investigations showed no clinically relevant findings. A 12- leads ECG recording showed a sinus rhythm with a heart rate (HR) of 90 bpm. An inhalation dose of 400ug salbutamol has been administered to perform post bronchodilator (PBD) pulmonary function tests and collect spontaneously expectorated sputum. The patient’s cardiac electrical activity was monitored (by means of telemetry) before (baseline, BL) and after salbutamol administration. A pre-bronchodilator (Pre-BD) telemetric recording showed episodes of asymptomatic tachycardia with fluctuating resting heart rate between 79 to 117 bpm, without ECG signs of myocardial ischemia. At 45-60 minutes after salbutamol administration, the telemetric recordings showed sinus tachycardia with resting HR fluctuating between 97 to 141 bpm and ECG manifestation of tachycardia-induced myocardial ischemia (Figures 1 and 2). 3 to 4 hours post salbutamol the HR returned to BL, and the patient has been discharged after ensuring her safety. Thyroid function tests have been performed to verify the aetiology of the tachycardia. These demonstrated normal results. Re-challenge tests have been performed on other visit. These confirmed the salbutamol associated cardiac effect.
Discussion & Conclusion: B2 agonist should be administered with caution to tachycardic CF patients. This may imply salbutamol dose adjustment and requires monitoring with cardiac telemetry. This might avoid unwanted cardiovascular adverse effects such as MI. This line of reasoning is substantiated by the increased likelihood of CF- associated cardiac CFTR chloride channel abnormality that results in deficient CFTR- induced cardiac ischaemic preconditioning and therefore increase the risk for ischemic heart disease in CF patients, particularly in the older age group with pre-existing decreased cardiac reserve.
Keynote Forum
Lynda Spelman
Veracity Clinical Research, Australia
Keynote: AWI002: Trials, tribulations and the value of persisting with non-drug related clinical trials – superfine merino wool base layer garments in atopic dermatitis
Time : 11:15-11:45

Biography:
Lynda Spelman is the Principal Investigator of Veracity Clinical Research. As a graduate of the University of Queensland and Fellow of the Australasian College of Dermatologists, she has worked as a Specialist Dermatologist in Australia for more than 20 years. She has an extensive 25 year history of involvement in clinical research, having conducted studies in trials for a wide range of dermatological conditions, including atopic dermatitis/eczema, chronic plaque psoriasis, psoriatic nail disease, palmoplantar psoriasis, hidradenitis suppurativa, seborrhoeic keratosis, and superficial and nodular basal cell carcinoma
Abstract:
In the pursuit of finding an alternative to the three traditional means of treating Atopic Dermatitis (AD) (moisturisers, topical corticosteroids, and systemic immunosuppressants), my team undertook a study that looked at the use of Superfine Merino Wool Base Layer Garments (SMWBLG) as a therapeutic intervention in the management of atopic dermatitis in children and young adults. This was an investigator lead study which received support from Australian Wool Innovation in the form of partial investigator funding and supply of the therapeutic intervention. Because the use of wool was a foreign concept in the management of patients with AD, we had to overcome many unexpected challenges and obstacles, the main one being the urban legend that wool causes allergic contact dermatitis. This was a HREC approved, proof of concept study and the second of its kind in collaboration with AWI. After proving the significant therapeutic effect that the garments had on adults, this study aimed to confirm the same was true in children and young adults. We continued to experience a number of hurdles in both completing the study and publishing this novel concept. Our team persisted through the study investigation phase and completed it with the successful proof of concept. Our study found that overall there was a significant decrease in AD extent, severity and subjective symptoms during the garment intervention phase compared to baseline and some significant effects on intensity and subjective symptoms even after the garments were removed, revealing an extended therapeutic effect. Despite evidence based research, editors commented on the relevance and significance of the use of this reusable, sustainable, effective intervention that has no systemic implications in this young population group with a chronic disease. The process of conducting this study has shown us that we must challenge current thinking, stay true to beliefs and gut feelings that practical interventions you see working each day are worth formally exploring
Keynote Forum
Lynda Spelman
Veracity Clinical Research, Australia
Keynote: AWI002: Trials, tribulations and the value of persisting with non-drug related clinical trials – superfine merino wool base layer garments in atopic dermatitis
Time : 11:15-11:45

Biography:
Lynda Spelman is the Principal Investigator of Veracity Clinical Research. As a graduate of the University of Queensland and Fellow of the Australasian College of Dermatologists, she has worked as a Specialist Dermatologist in Australia for more than 20 years. She has an extensive 25 year history of involvement in clinical research, having conducted studies in trials for a wide range of dermatological conditions, including atopic dermatitis/eczema, chronic plaque psoriasis, psoriatic nail disease, palmoplantar psoriasis, hidradenitis suppurativa, seborrhoeic keratosis, and superficial and nodular basal cell carcinoma
Abstract:
In the pursuit of finding an alternative to the three traditional means of treating Atopic Dermatitis (AD) (moisturisers, topical corticosteroids, and systemic immunosuppressants), my team undertook a study that looked at the use of Superfine Merino Wool Base Layer Garments (SMWBLG) as a therapeutic intervention in the management of atopic dermatitis in children and young adults. This was an investigator lead study which received support from Australian Wool Innovation in the form of partial investigator funding and supply of the therapeutic intervention. Because the use of wool was a foreign concept in the management of patients with AD, we had to overcome many unexpected challenges and obstacles, the main one being the urban legend that wool causes allergic contact dermatitis. This was a HREC approved, proof of concept study and the second of its kind in collaboration with AWI. After proving the significant therapeutic effect that the garments had on adults, this study aimed to confirm the same was true in children and young adults. We continued to experience a number of hurdles in both completing the study and publishing this novel concept. Our team persisted through the study investigation phase and completed it with the successful proof of concept. Our study found that overall there was a significant decrease in AD extent, severity and subjective symptoms during the garment intervention phase compared to baseline and some significant effects on intensity and subjective symptoms even after the garments were removed, revealing an extended therapeutic effect. Despite evidence based research, editors commented on the relevance and significance of the use of this reusable, sustainable, effective intervention that has no systemic implications in this young population group with a chronic disease. The process of conducting this study has shown us that we must challenge current thinking, stay true to beliefs and gut feelings that practical interventions you see working each day are worth formally exploring
- Clinical Research | Innovations in Pre-clinical Research | Design of Clinical Studies and Trials | Clinical Data Management and Statistics | Stem Cell & Oncology Clinical Research | Innovations in Clinical Trials | Clinical Trials on Different Diseases | Clinical Trials in Developing Countries | Bioethics and Quality Regulation
Location: Redwood Suite A

Chair
Nicole Powell
Statistics and Data Corporation, USA

Co-Chair
Tanguy Jerome
Nestle Research Center, Switzerland
Session Introduction
Tanguy Jerome
Nestle Research Center, Switzerland
Title: Challenges in clinical nutritional research: How adaptive design can help?
Time : 11:50-12:40

Biography:
Tanguy Jerome has been working in the field of Biostatistics for 10 years in R&D entities of international food companies. Over the years, he contributed to the development of the statistical thinking and clinical trials within the food industry. During the last decade, his methodological research was mainly focused on advanced modeling (mixture modeling, frailty model and latent process model) which has led to yearly contribution during international statistical congresses. Recently, his field of interest focused on the implementation and development of adaptive trials in nutritional research
Abstract:
Human studies are essential to obtain evidence of beneficial effects of nutritional interventions on health. Clinical nutritional development plans include a range from small pilot trials to well-designed confirmatory studies, following good clinical practices. Nutrition interventions are generally safe compared to pharmaceutical compounds. Regulatory guidance limits the benefits of nutrition to the maintenance of normal function or the reduction of accepted risk factors. Therefore, nutritional trials do not necessarily follow the same development process as pharmaceutical products. The objectives focus more on maintaining health and preventing risk factors for disease rather than showing efficacy in disease conditions. Clinical trials in the nutritional field have specific challenges to overcome in terms of population, intervention, design and methodology. Additional challenges are the lack of learning phases, small beneficial effects, high heterogeneity of the responses and difficulties in finding appropriate endpoints. In recent years, the use of adaptive design methods in clinical trials has received much attention due to its flexibility and efficiency in clinical development. In practice, adaptive design may provide the opportunity to modify certain aspects of the trial design while the study is still ongoing, without violating the quality and the integrity of the data. However, introducing more flexibility may come with many difficulties: major adaptations of on-going trials may result in designs unable to address the original scientific/medical questions. In addition, fundamental differences between pharmaceutical and nutritional research potentially trigger limitations but also opportunities for the application of such innovative design. The aim of this work is to highlight specificities and challenges in nutritional clinical trials, review the concepts of adaptive design and then address the advantages, limitations, and feasibility of commonly considered adaptive designs. Statistical, clinical, operational and regulatory aspects of implementing adaptive designs will be discussed
Lynda Spelman
Veracity Clinical Research, Australia
Title: National dermatology radiation oncology registry: A human research registry to collect information from patients referred to radiation oncologists for the management of non-melanoma and melanoma skin cancer and difficult to manage inflammatory skin diseases through radiotherapy
Time : 12:40-13:05

Biography:
Lynda Spelman is the Principal Investigator of Veracity Clinical Research. As a graduate of the University of Queensland and Fellow of the Australasian College of Dermatologists, she has worked as a Specialist Dermatologist in Australia for more than 20 years. She has an extensive 25 year history of involvement in clinical research, having conducted studies in trials for a wide range of dermatological conditions, including atopic dermatitis/eczema, chronic plaque psoriasis, psoriatic nail disease, palmoplantar psoriasis, seborrheic keratosis, and superficial and nodular basal cell carcinoma and other skin cancers
Abstract:
Rationale: Radiotherapy is the use of ionizing radiation to treat cancer and related diseases and has often been utilized as a treatment option for skin cancers. The registry will be used to analyse and assist in the development of best clinical practice for dermatologists and radiation oncologists.
Aim: To objectively measure baseline characteristics, e.g., demographic, medical history, treatment schedule, outcomes and follow-up of patients undergoing radiotherapy for skin cancers.
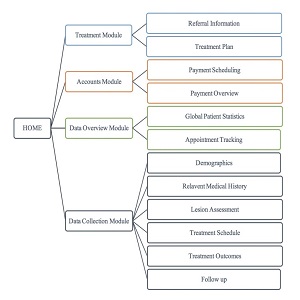
Registry Design: The registry design provides an accurate, non-interventionist approach to documenting patient outcomes and information. The Online Secure Platform (OSP) has four key modules (see Figure 1).
Study Schedule: Data collection will take place at the Registry enrolment by Referring Doctors (RD), treatment planning with Treating Radiation Oncologists (TRO), during treatment visit, and approximately every 3 months for the first 12 months and then 6 monthly thereafter.
Data Security & Handling: Data from the registry will be stored through the OSP on an Australian server accessible by an encrypted online portal, accessible only by the direct RD and TROs and their staff.
Monitoring Plan: A trained representative will monitor registry data entry, patient visit status, accuracy of information and missing data. All changed information, including the date and persons performing the corrections, will be available via the audit trail.
Multi-site Research Logistics: This study involves a large number of sites both by RDs and TROs. RDs are located in over 20 sites in Australia. TROs are located at Genesis Cancer Care sites across Australia.
Statistical Analysis: Registry reports will be written detailing the objectives, methodology, results, and conclusions. Bellberry human ethics will be consulted in this process.
Significance: The creation of a functional registry is not a straight forward process. A detailed, obsessional, establishment is necessary to ensure that the future use of its data is relevant
Stefan Franzén
Centre of Registers, Sweden
Title: Evaluation of swedish integrated electronic health records and register health care data to serve as a reference population in severe asthma clinical trials: The PACEHR study
Time : 13:45-14:10

Biography:
Stefan Franzén completed his PhD in Mathematical Statistics and after spending more than ten years as a Statistician in the pharmaceutical industry, he currently involved with epidemiological research in diabetes at the public sector Swedish National Diabetes Registry
Abstract:
Objectives: In the development of new drugs for severe asthma, it is a challenge from an ethical point of view to randomize severe asthma patients to placebo, and to obtain long-term safety data due to discontinuations. The aim of this study was to evaluate the feasibility of using electronic health record (EHR) data to create a real-world reference population of uncontrolled asthmatic patients to replace or supplement the concurrent control/placebo group in long-term studies of asthma.
Methods: EHR data from 36 primary care centres and a University hospital in Sweden were linked to Swedish mandatory health registers (2005–2013), creating a population covering 33 890 asthma patients, including data on co-morbidities, risk factors and laboratory/respiratory measurements. A severe asthma EHR reference cohort was established. We used logistic regression to estimate the propensity score (probability) of each RCT or EHR patient existing in the EHR cohort given their covariates.
Results: We created an EHR-derived reference cohort of 240 patients, matching the placebo group (N=151) in an RCT of severe asthma. The exacerbation rate during follow-up in the EHR study population was 1.24 (weighted) compared to 0.9 in the RCT placebo group. Patients in the EHR cohort were of similar age as in the RCT placebo group, 50.6 years versus 50.1 years; had slightly higher body mass index 27.0 kg/m2 versus 27.3 kg/m2; and consisted of 40% versus 34% males.
Conclusions: The results indicate that EHRs provide an opportunity to supplement, and in the future potentially replace, the control group in RCTs of severe diseases.
Nicole Powell
Statistics and Data Corporation, USA
Title: The importance of choosing the right partners and technologies to create the most efficient clinical trials
Time : 14:10-14:35

Biography:
Nicole Powell has spent over 12 years in the industry building effective partnerships mostly focused on CRO’s and sponsor relationships. She has a passion for creating specialized experiences for sponsors that enable the clinical trial process to run efficient and smooth. Her ability to combine real world trial experience with the many changes in the industry, over the past decade and beyond, is top notch and provides sponsors with an out of the box solution to create effective trials. Most of her concentration has been focused on evaluating partner relationships, new technologies, and the importance of correct data management and statistical application in trials. She currently plays a pivotal role in the International Business Development and Sales team at Statistics and Data Corporation (SDC) with offices in Boston and Phoenix
Abstract:
Statement of the Problem: Costs and regulatory requirements in our industry are ever increasing and many Sponsors are not aware of how to choose the correct service partner to fit their needs when costs and resources are such a large constraint. Larger CRO’s aren’t inherently set to work with smaller/mid-size Sponsors and therefore don’t have the complete flexibility to create customized solutions to meet the very specific and individualized needs of, for example a virtual Sponsor. Emphasis needs to be placed on statistical consulting from the beginning of the study. Additionally, there are many new technology solutions being pushed upon Sponsors that it is sometimes hard to get past the buzz words and truly evaluate what you need to create an efficient clinical trial.
Methodology & Theoretical Orientation: Sponsors need to decide what existing internal capabilities they have, along with the experience their internal employees bring. They also need to understand the technologies their team is familiar with good and bad experiences and pull from the internal resources they have before exploring partnering options. Once that is established they must look outward to find a partner which shares the same values, service approach and ability to meet their unique needs.
Findings: An increased importance placed on statistics early on in the protocol process can help save time as the trial continues, and can help avoid potential introductory questions by Regulatory Agencies. A chosen partner needs to compliment internal services, have open communication plans put in place and most importantly have long standing relationships with other strategic partners to meet all of the Sponsors needs with less work from the Sponsor. An already established strategic partnership network of specialty providers creates a seamless experience for the Sponsor and a long-standing relationship to develop and execute an efficient trial
Guido C Mora
Universidad Andres Bello, Chile
Title: Pseudogenes contribute to Salmonella typhi pathogenesis in humans
Time : 14:35-15:00

Biography:
Guido C. Mora is working as an full professor, microbiology, School of Medicine, Universidad Andrés Bello, and adjunct professor San Diego state university previously he was the dean of ecology and natural resources 2007-2011, In 2009 he was the ambassador, American society for microbiology for Chile, Argentina, Uruguay y Paraguay his research includes the clinical research, microbiology, biochemistry, he participated in various national & international conferences all around the world. He published many articles in the reputed journals
Abstract:
Salmonella enterica serovars, in spite of sharing a highly similar genomic content, exhibit a fundamental difference in the range of host they infect and in the disease they cause. Some serovars such as Salmonella typhimurium, are called generalist because they infect several mammalian hosts and other, such as Salmonella typhi, are called specialist because their target is one and only host, in this case the human being. S. typhi causes typhoid a systemic infection which is a potentially lethal disease in contrast to S. typhimurium causing a self-limited gastroenteritis, rarely lethal. The evolution of S. typhi towards a strictly human pathogen may have arisen by acquisition of new functions, loss of functions, or by a combination of both mechanisms. It is understandable that the gain of function can provide a selective advantage to a bacterium in a particular environment. Nevertheless, the loss of functions also can drive pathogen evolution, either by eliminating a function that reduces growth in a particular niche or by the energy savings associated with eliminating a function that is no longer needed. Therefore, our working hypothesis is that the difference in the disease caused in humans by S. typhi and S. typhimurium is due to, at least in part, by the gain and/or loss of functions. This presentation will be dedicated to show that pseudogenization or the erosion of some genes may modify bacterial functions leading S. typhi to a better fit in its host because the study of S. typhi pathogenic mechanisms are hampered by the nature of its host. Our approach is to work with culture cell lines. This presentation will be dedicated to show the effect of two interesting pseudogenes, sseJ and marT. In S. typhimurium both are active genes encoding SseJ and MarT. The former corresponds to an acyltransferase/lipase that participates in Salmonella containing vacuole biogenesis in human epithelial cell lines and is needed for full virulence of S. typhimurium. On the contrary, our work suggest that sseJ inactivation in S. typhi has important role in the systemic infection. The latter is a transcriptional regulator as part of marT-fidL operon located at SPI-3. Our work shows that S. typhi expressing S. typhimurium marT-fidL exhibited an increased accumulation of reactive oxygen species (ROS), leading to a decreased survival in presence of H2O2 that also affected survival within macrophage-like cells. The genes loss included in this presentation are supporting the notion that pseudogenization in S. typhi is to gain a better fit and virulence in its host
Mona Dawood
Centre International de Développement Pharmaceutique, Mauritius
Title: Conducting a clinical trial in Mauritius: Legal, regulatory and ethical frameworks
Time : 15:00-15:25

Biography:
Mona Dawood is Head of the Pharmaceutical Operations & Regulatory Affairs for CIDP group. She completed BSc (Hons) in Biomedical Chemistry from University of Warwick (UK) and a Diploma in CRA from Sup-Santé, Paris, France. She joined CIDP seven years ago and has acquired a significant expertise in specific monitoring in various therapeutic areas (oncology, diabetes and pediatrics). She has also an excellent knowledge of the European & Asian regulations, regulatory aspects, claims validations, pharmacovigilance
Abstract:
Since 1980’s, clinical research in Mauritius has been limited to small observational and population-based studies, often with a strictly national relevance. However, the establishment of a legal framework for clinical trials in 2011 boosted the interest of sponsors to conduct interventional trials. Since 2011, Mauritius has seen a major development in its life sciences and research sector with the development of clinical research organizations, set-up of clinical research units in hospitals, and the growing training given to local investigators and clinical staffs. Indeed, the Clinical Trials Act provides for the setting up of a Clinical Research Regulatory Council (CRRC) responsible for the regulation and control of trial licenses being issued, an Ethics Committee (EC) to advise the CRRC regarding welfare, safety, health and protection of human subjects participating in clinical trials, and a Pharmacovigilance Committee (PC) to monitor all clinical trials being performed and ensure Good Clinical Practice (GCP). We propose to describe in this poster the legal framework for clinical research, and the regulatory process for conducting a clinical study in Mauritius. The objective of this presentation is to confront international guidelines and regulations for conducting a clinical trial (Good Clinical Practices, FDA Guidelines) to the Mauritian dispositions and laws. A second objective is to question the ethics of research in Mauritius, in the lights of concerns raised by recent publications about clinical research in developing countries.
Shuo Chen
Institute of hematology and Blood Diseases Hospital – CAS, China
Title: The possibility of the use of single Institutional Review Board (IRB) for multi-site research in China
Time : 15:25-15:50

Biography:
Lijun Liu has his expertise in the administrative work of clinical trial. He has been working for the clinical trials in the hospital. He was also the secretary of the ethic committee in hospital, who did a lot of pioneering work for the initiation of ethic committee. He is one of the CFDA (China Food and Drug Administration) inspectors in China and helped inspection work several times
Abstract:
With the rapid development of biomedicine in recent years, multi-site research has become the world trend; as a result, there is no other way but to apply for multi-IRB review for the commencement of the trial. The duplicate IRB review by each site increases administrative burdens and cost for sponsors and brings no substantive good to the human subject protection. In October 2016, National Health and Family Planning Commission in China issued Guideline on the IRB review on the biomedical research involving human subjects. It mentions in order to keep the consistency and efficiency of multi-center research, the IRB in each site can establish a collaborative review mechanism, which means the lead site reviews the protocol and rely site reviews its feasibility. In June 2016, the National Institutes of Health (NIH) issued new guidance on single-IRB review of multicenter studies, which brings a new era in multicenter studies
Chay-Hoon Tan
National University of Singapore, Singapore
Title: Usage of herbal medicines in older adults in Singapore
Time : 15:50-16:15

Biography:
Chay-Hoon Tan is the Clinical Associate Professor in Department of Pharmacology, National University of Singapore and Consultant Psychiatrist in the Department of Psychological Medicine, National University Hospital
Abstract:
Introduction: Herbal medicines (HM) are a complementary and alternative medicine (CAM) modality that will likely play greater roles in older adults’ care. HM’s oral nature coupled with age-related pharmacological changes, comorbidities and poly-pharmacy put older adults at risk of drug-herb interactions, adverse reactions and hospitalizations. However, current studies focus largely on CAM use in the general population.
Objective: To investigate the utilization patterns and factors associated with HM use of older adults in Singapore.
Methods: A cross-sectional study of subjects from association between diet and health status study (project 10-517). Sociodemographic and health variables were collected from project 10-517 and telephone interviews regarding HM utilization were conducted on corresponding subjects. Inclusion criteria of subjects were: no intervention study participation; written consent for follow-up and; verbal consent. Exclusion criteria included inability to contact/interview after ≥3 attempts and; inability to speak on phone.
Results: 509 subjects were interviewed. Subjects’ mean age was 67±6 years old. 12 month HM prevalence was 60.1%. Top 3 HM included American ginseng (n=161, 52.6%), wolfberry (n=104, 34.0%) and Codonopsis (n=69, 22.5%). They were mainly used for health improvement with high satisfaction levels. Sociodemographic predictor variables included gender (female OR:3.199, CI:2.021-5.065), age (≥76 years old, OR:0.444, CI:0.198–0.997) and high social activities level (high, OR:1.964, CI:1.012–3.813). Health predictor factors included family history of dementia (OR:3.157, CI:1.036–9.618), improvement of self-rated health (OR:1.799, CI:1.020–3.173) and specific chronic conditions like arthritis (OR:1.994,CI:1.039-3.823) and cancer (OR:0.367,CI:0.148-0.911).
Conclusion: HM utilization patterns, sociodemographic and health predictor variables found in this study can aid healthcare professionals to identify potential/current HM users and initiate individualized education/consultation on safe HM use among older adults.
Yize Li
Fourth Military Medical University, China
Title: LPS induces HUVEC angiogenesis in vitro through miR-146a-mediated TGF-β1 inhibition
Time : 16:15-16:40

Biography:
Yize Li is Doctor at Xijing Hospital, Fourth Military Medical University. She completed her Bachelor degree of Clinical Medicine at West China Medical Center Sichuan University and Master and PhD degree at Fourth Military Medical University. She is skilled in treatment of breast cancer. She focus her research in lncRNA and microRNA and explore the mechanisms of trastuzumab resistance in breast cancer and endothelial cells angiogenesis.
Abstract:
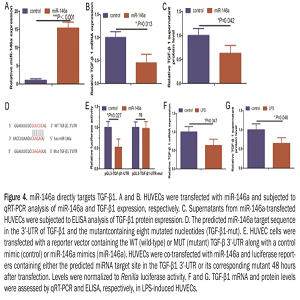
Angiogenesis is an essential process for tissue growth and embryo development. However, inflammation, abnormal wound healing, vascular diseases, and tumor development and progression can result from inappropriate angiogenesis. Lipopolysaccharide (LPS) can activate various cells and alter endothelium function and angiogenesis. This study investigated the underlying molecular events involved in LPS-induced angiogenesis and revealed a novel strategy for controlling abnormal angiogenesis. LPS treatment promoted wound healing and tube formation in human umbilical vein endothelial cell (HUVEC) cultures and induced their expression of miR-146a. miR-146a was previously shown to regulate angiogenesis in HUVECs. Knockdown of miR-146a expression antagonized LPS-induced angiogenesis in vitro. Moreover, bioinformatics analyses predicted TGF-β1 as a target gene for miR-146a, which was confirmed by a luciferase reporter assay. Expression of miR-146a in HUVECs resulted in down-regulation of TGF-β1 in HUVECs, whereas a miR-146a inhibitor upregulated the expression of TGF-β1 and TGF-β1 downstream proteins, such as phosphoraylation-Smad2 and plasminogen activator inhibitor type 1 (PAI-1). Furthermore, the TGF-β1 signaling inhibitor SB431542 impaired the ability of miR-146a knockdown to suppress LPS-induced angiogenesis. Thus, LPS-induced angiogenesis of HUVECs functions through miR-146a up-regulation and TGF-β1 inhibition. This study suggests that knockdown of miR-146a could activate TGF-β1 signaling to inhibit angiogenesis as a potential therapy for angiogenesis-related diseases.
Khalid Abou Farha
Clinical Pharmacology Research Centre, UK
Title: Muller's manoeuvre, potential non-pharmacological bronchodilator relevance in clinical trials
Time : 16:55-17:20

Biography:
Khalid Abou Farha is medical director and clinical pharmacologist of Celeron he brings 15 years of clinical research experience to this role, spanning a wide range of therapeutic areas, with a particular focus in oncology, vaccines, cancer immunotherapy, personalized medicine and autoimmune disorders. He has extensive experience in building and managing global teams, leading the development of Clinical Research drugs and biologics
Abstract:
Background: Many respiratory clinical trials include sputum induction using hypertonic saline to evaluate airway inflammation and monitor treatment response. Inhalation of hypertonic saline may cause bronchoconstriction that necessitate discontinuation of the procedure and administration of bronchodilator therapy such as salbutamol. Müller’s manoeuvr is a technique used in the diagnosis of sleep apnoea syndrome. The associated physiological response includes increased sympathetic flow and withdrawal of parasympathetic activity. Given that, we hypothesized that this manoeuvre might exhibit a bronchodilator effect and therefore could be beneficial in patients with bronchospasm.
Patients & Methods: The potential bronchodilator effect of Müller’s manoeuvre has been assessed in 10 patients with moderate to severe COPD, who underwent sputum induction procedure, and 9 healthy non-smoking subjects. The latter group has been included to explore the airway response to the manoeuvre in healthy individuals. The manoeuvre (voluntary inspiration against closed nose and mouth) was explained to all participants. Pre- and post- manoeuvre assessment of 4 spirometry parameters, FVC, FEV1, PEF and PEF25-75, has been performed. Assessment of post manoeuvre changes has been performed over 25 minutes and divided into 4 intervals of 5 minutes, A to D.
Results & Conclusion: Sputum induction resulted in asymptomatic bronchoconstriction with >20% decline in FEV1, FVC and PEF, compared to baseline (BL). The use of Muller’s manoeuvre led to an increase in the means of declined values (P<0.01%). Figure 1 depicts s Müller’s manoeuvre - induced changes in the means of assessed parameters, compared to post sputum induction (PSI). FEV1 of all 10 patients returned to ≥ 90% of their BL without the use of pharmacological bronchodilator. In healthy volunteers the average changes due to Müller’s manoeuvre relative to baseline values were + 101 ml (+ 2.5%), + 118 (+ 3.5%) and 170 ml (+ 4.7%) respectively in FVC, FEV1, and PEF. Muller’s manoeuvre showed a potential bronchodilator effect and could reverse PSI bronchoconstriction in 10 COPD patients. The use of this manoeuvre obviated the requirement to use bronchodilator therapy in all 10 patients, and therefore might be potential alternative to B2 agonists in COPD cardiac risk patients participating in clinical trials. Further investigations to confirm this finding are still warranted.
Shahryar Eghtesadi
Azad University Science and Research Branch, Iran
Title: Effects of hesperidin supplementation on weight, glycemic control, lipid proï¬le and inflammatory factors in patients with type 2 diabetes: A randomized, double-blind and clinical trial placebo-controlled
Time : 17:20-17:45

Biography:
Shahryar Eghtesadi received Bachelor degree in Nutrition Science and Food Chemistry in 1975, from Shahid Beheshti University of Medical Sciences, Tehran; MSPH degree in Nutrition, 1977, from Tehran University of Medical Sciences, Tehran and PhD from University of California at Davis (UCD), USA, in Nutrition (1985). He served as Visiting Scientist in USDA Human Nutrition Research Center on Aging (HNRCA), Boston, USA (1994-1995); Full professor of Tabriz, Iran and Tehran Universities of Medical Sciences and currently serves as Professor of Azad University, Science & Research Branch. He served as Associate Dean and Dean of School of Public Health & Nutrition and School of Public Health of Tabriz and Iran University of Medical Sciences respectively. He was selected as distinguished Professor and Scientist
Abstract:
This study was conducted to investigate the effects of hesperidin (a common constituent of citrus fruits) supplementation on weight, indices of glycemic control, insulin resistance, lipid proï¬le, and inflammatory markers in patients with type 2 diabetes. 45 patients participated in this randomized, double-blind controlled clinical trial who were randomly assigned to 2 intervention and control groups. Participants consumed either 500 mg/d pure hesperidin supplement or placebo in the intervention and control groups for eight weeks, respectively. Hesperidin supplementation led to significant decrease in fasting blood glucose (FBG) and glycated hemoglobin (HbA1c) (p=0.041 and p=0.028, respectively). A significant increase in serum insulin (p=0.018) and decrease in total cholesterol (p=0.049) were also observed in the hesperidin group, whereas no signiï¬cant changes occurred in the placebo group. Inflammatory factors, high-sensitivity C-reactive protein (hs-CRP) and interleukin-6 (IL-6) were not signiï¬cantly changed in the hesperidin group compared to the control group. In conclusion hesperidin supplementation lowered the plasma level of total cholesterol and improved glycemic control and insulin resistance in patients with type 2 diabetes
- Biomedical Devices Clinical Research | Clinical Trials in Developing Countries | Clinical Trials in Developing Countries | Design of Clinical Studies and Trials | Clinical Trials on Different Diseases
Location: Redwood Suite A

Chair
Victor George Aeby
East Carolina University, USA

Co-Chair
Tracy Carpenter-Aeby
East Carolina University, USA
Session Introduction
Tracy Carpenter-Aeby
East Carolina University, USA
Title: The evolution of psychopathology in social work
Time : 11:45-12:35

Biography:
Tracy Carpenter-Aeby is a professor, clinical social worker, supervisor and an intervention researcher. She is devoted to integrating research and teaching into professional practice to improve the lives of clients. She is also dedicated to mentoring students and shepherding professionals so that they may contribute to changes in policy and improvements in practice for the wellbeing of clients. Her intentional, thoughtful, strategic study of social injustices through the lens of intervention research promises transformational changes in the quality of life for clients by building on evidence-based practice. Working with students who enter the social work profession and, often, practice in underserved, rural communities gratifies her. Through her research, she has helped to alter the fabric of those communities by preparing practitioners armed with evidence-based practice and trained to facilitate intentional, deliberate, life-affirming change with vulnerable clients
Abstract:
The term psychopathology dates back to the ancient civilization of Hippocrates and Aristotle. Yet, it did not gain wide acceptance in practice until advocates like Freud, Kraepelin, and Meyer applied it to practice with clients. The acceptance of psychopathology in practice has been slow and tumultuous. The National Association of Social Workers stated that a vast majority of providers of mental health services in the United States are social workers. For mental health practice, the most widely used assessment system has been and is the American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Health, DSM. About 20% of Americans have experienced psychiatric disorders, and this figure is expected to be increasing. Despite one’s career path within the field of social work, practitioners are more than likely going to encounter clients with a mental illness. Thus, those who work with the mentally will need to learn how to decipher the DSM format
Sarah Moran
Mater Misericordiae University Hospital, Dublin
Title: Intravitreal bevacizumab compared with diode laser in stage 3 posterior retinopathy of prematurity: A 5 year follow up
Time : 12:35-13:00

Biography:
Sarah Moran is a Specialist Registrar in Ophthalmic Surgery in Ireland with numerous published research articles and multiple national and international presentations in the area of ophthalmic surgery
Abstract:
Introduction: Retinopathy of prematurity (ROP) is a potentially blinding eye disorder occurring in premature infants. Vascular endothelial growth factor (VEGF) is central to its pathogenesis. The use of anti-VEGF agents has become an alternative treatment in some babies.
Methodology: We concluded a prospective randomized trial in 15 babies, gestation 24-26 weeks and birth weight 500-1000 grams. We compared diode laser in one eye to Intravitreal bevacizumab in the fellow eye in stage 3 plus disease in zone 1 and posterior zone 2 of the retina. We recorded regression, recurrence, vision, refraction and toxicity.
Findings: The bevacizumab treated eyes showed rapid regression of ROP with resolution of plus disease and flattening of the ridge at 48 hours post injection. Retinopathy of prematurity recurred in four eyes of babies treated with intravitreal bevacizumab. These were successfully treated with laser or a repeat injection of bevacizumab. At 5 year follow up, the bevacizumab treated eyes are less myopic compared with the diode laser treated eyes. All 15 infants had magnetic resonance imaging (MRI) of brain performed, and there were no changes or adverse effects that could be attributed to the bevacizumab therapy.
Conclusion: Bevacizumab is an effective treatment for halting ROP, and appears to be safe in a dose of 1.65mg in 0.1ml. Refractive outcomes are more favorable in bevacizumab treated eyes. There were no ocular or systemic adverse effects attributed to bevacizumab therapy. There was a recurrence of ROP in four eyes and they required repeat bevacizumab or laser. Eyes treated with bevacizumab require follow up to sixty weeks post menstrual age. At five years we encountered no ocular or systemic side effects
Louise Fitzgerald
Our Lady’s Children’s Hospital Crumlin, Ireland
Title: Encouraging oral analgesia in the fasted child attending a trauma review clinic
Time : 14:00-14:25

Biography:
Abstract:
Background: Primary presentation of soft tissue injuries is often to units such as emergency departments and general practitioners. Following consultation with an on call plastic surgery team, a review appointment is commonly planned for the following morning for injuries that do not threaten a patient’s systemic health or compromise perfusion. Patients are requested to arrive at the Trauma Review Clinic fasting, so as to allow general anaesthesia to be undertaken. Analgesia should be administered to pediatric patients prior to assessment of wounds. The fasting status of a potentially pre-operative patient should not deter parents or referring clinicians from recommending and administrating oral analgesia before review.
Aim: Investigate the administration of analgesia in pediatric patients attending a trauma review clinic with amendment of the referral process to optimize pain management.
Method: A prospective closed loop audit of two consecutive groups of 50 patients attending a trauma review clinic was performed. Data collected included analgesia type and quantity, fasting status, reason for withholding analgesia, and pain scores during and after wound inspection. Intervention was performed using published advice for GPs, emergency department education and emails to all clinicians accepting referrals.
Results: Of the pre-intervention patients, 94% (47/50) had no analgesia and 68% (34/50) of these were not advised to take analgesia by referring clinician. Of the post-intervention patients, 64% (32/50) had no analgesia of which 32% (16/50) were not advised to take analgesia by referring clinician. Pain scores were similar for both groups during wound assessment; however post assessment average pain scores were significantly reduced in the post intervention group (2.3/5 pre intervention, 1.4/5 post intervention).
Conclusion: Despite published guidelines regarding fasting times, children are not administered oral analgesia when attending for trauma wound inspection. Simple clinician education can improve administration rates resulting in reduced pain scores
Wael Nasser
Baruch-Padeh Poriya Medical Center, Israel
Title: The relationship between urinary tract infection and vesicoureteral reflux in children from Eastern Galilee Region-Israel
Time : 14:25-14:50

Biography:
Wael Nasser is the Head of Pediatric Nephrology department at Baruch Padeh Poriya Medical Center. He is responsible for pediatric nephrology at Faculty of Medicine, Member of the Israeli Association of Nephrology, Israel Society of Pediatrics, and Israel Society of Hypertension. He has experience in research, evaluation, teaching and administration both in hospital and education institutions
Abstract:
Background & Aim: The incidence of vesicoureteral (VUR) in the general population is less than 1%. Reflux carries an increased risk of pyelonephritis and renal failure. The purpose of the current study was to check whether the voiding cystourethrography (VCUG) test is necessary in most cases of upper urinary tract infection (UTI) in children, and to investigate if there is a correlation between the presence of VUR and urinary tract infection, or the reflux is a random finding that does not require monitoring. We studied the correlation with various markers of inflammation, renal ultrasound findings (US), (DMSA) nuclear renal scans findings, and the clinical presentation.
Methods: The current study was based on tests taken at the Poriya Medical Center in Tiberius, between 2009 to 2013. The study includes 88 children up to 2 years that were asked for VCUG. All the children had symptoms of UTI. The children had laboratory tests (blood, urine, culture), then renal US. Older children did the voiding test after doing DMSA. DMSA was done 3-5 months after the acute UTI.
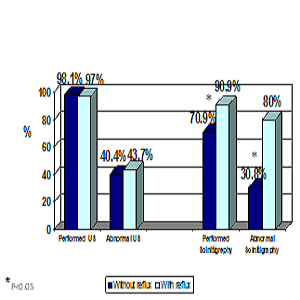
Results: Approximately 40% (37.5%) of patients suffered from reflux: 66.7% with incorrect DMSA (filling defect/scar) had reflux. Only 18.2 % of children with normal DMSA results had reflux. High inflammation factors levels (CRP, ESR) were found in all subjects with reflux, and only 52.6% of cases with positive reflux had low lymphocytes levels compared with only 14.3% in patients without reflux.
Conclusions: VCUG is not required in most cases of upper UTI. Suitable replacement test can be done by renal DMSA, which has also an added value of detecting a potential renal damage (scars). High levels of inflammation factors, erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP) strongly suggest a possible existence of reflux. In contrast, lower levels deny the existence of reflux in children with upper UTI.
Hashimita Sanyal
University College Dublin, Ireland
Title: Effect of estradiol and progesterone on ovine amniotic epithelial cells
Time : 14:50-15:15

Biography:
Hashimita Sanyal is pursuing her PhD and has expertise in Nano-biotechnology. Prior to working with stem cells, she had worked on projects like bruxism, biosensors for detecting the bacteria Legionella, nanoparticles for drug delivery and more. With her current research venture in stem cells, she wants to explore the effect of the two important female sex hormones on amniotic epithelial cells
Abstract:
Amniotic-derived epithelial cells (AECs), an emerging source of fetal stem cells, have recently attracted the attention of researchers for their great regenerative potential. Because of their fetal origin, these cells exhibit elevated proliferation rates and plasticity, as well as, immune tolerance and anti-inflammatory properties making AECs suitable for both allogenic and xenogenic transplantation. Recently, it was demonstrated that P4 supplementation is crucial to preserve epithelial phenotype and to enhance biological properties in expanded ovine AEC (oAECs), but to date no information are present on the effects of prolonged steroid exposition on oAECs. Starting from these premise, this study was designed to clarify the Estradiol (E2) and Progesterone (P4) steroid different concentrations (12, 5 and 25 uM) effects on oAECs proliferation, stemness and differentiation, by using histological, RT-qPCR, immunohistochemistry and cell counting analysis. Results indicate that oAECs proliferate with no differences between concentrations. P4 oAECs treated cells showed the epithelial shape with cytokeratin expression until third passage, while CTR and E2 treated cells down regulate cytokeratin and increase the αSMA expression protein after the first passage. Stemness genes OCT4, NANOG and SOX2 expressions were maintained higher in 12.5 µM E2, 25µM P4 and 25µM of both E2+P4 treated oAECs despite their progressive down-regulation in the CTR. Moreover, after 21 days of osteogenic induction in culture, steroids treated oAECs acquire chondrogenic-like morphology with alcian-blue positive stain despite the alizarin red stain positivity observed in no treated cells. In summary, prolonged steroids treatments can modify oAECs stemness properties and plasticity opening new prospective for the oAEC use in stem cell-based therapy in animal models
Lynnette Lyzwinski
University of Queensland, Australia
Title: The mindfulness app study for weight management, weight related behaviour, and stress in university students: A randomized controlled trial
Time : 15:15-15:40

Biography:
Lyzwinski L N completed her MPhil in Public Health from Cambridge University and; BA in Health Science from Simon Fraser University. She is pursuing her PhD in Medicine at University of Queensland with the Centre for Online Health and Department of Psychiatry. She is affiliated with the Princess Alexandria Hospital and the Royal Brisbane and Women’s Hospital. Her primary research interests include lifestyle medicine, RCTs, mHealth, e-health, obesity, and mindfulness
Abstract:
Background: University students are at risk gaining weight during their studies known as the Freshman 15. The prevailing literature has linked stress with engagement in maladaptive weight related behaviour in university students. Stress is highly prevalent in university students. Emerging research suggests that mindfulness may hold promise for assisting with stress as well as the adopting of healthy eating behaviour and weight management. One novel medium for delivering a weight loss intervention is through mobile health. Research suggests that mobile health interventions are effective for weight loss. Delivering a mindfulness-based intervention to students via a mobile medium may offer unique benefits including the possibility for students to learn mindfulness techniques at any time and place, something which is pertinent to them given their busy schedules.
Methods: A two arm randomized controlled trial will be conducted at the University of Queensland St Lucia and Herston campuses in students over the age of 18 years of age owning a smartphone who wish to lose weight. We will aim to recruit >50 and up to 115 participants. Students meeting eligibility criteria will be randomized to a mindfulness-based app or to a standard control group receiving an information leaflet on diet and physical activity guidelines. Changes assessed from baseline to follow-up at 3 months will be BMI, weight, dietary intake, physical activity, and stress. Feasibility and acceptability will also be assessed.
Discussion: This will be the first mobile mindfulness-based app trial in university students. If effective, this app intervention may hold potential for assisting students with managing key health issues pertinent to them.
Wenqing Feng
Shanghai Jiaotong University, China
Title: Design of clinical trials of effect on whether preserve Left Colonic Artery (LCA) on the prognosis of patients with laparoscopic resection of Inferior Mesenteric Artery (IMA) in rectal cancer
Time : 15:40-16:05

Biography:
Wenqing Feng completed her Master degree at Shanghai Jiaotong University School of Medicine. She studied under Professor Aiguo Lu (Director of Shanghai Minimally Invasive Surgical Center). Her research focuses on laparoscopic as well as robotic-assisted laparoscopic resection for rectal cancer, specifically on laparoscopic resection for mid and low rectal cancer
Abstract:
Incidence of colon cancer has reached the fourth among all cancers in China and there is an upward trend in the incidence of the disease, and the middle and low rectal cancer accounts for about 70% of all cancers. With the rapid development of science and technology, continuous improvement of surgical instruments, the application of laparoscopic radical resection for rectal cancer has become more and more mature and standardized. Treatment of inferior mesenteric artery is a key step in laparoscopic radical resection of rectal carcinoma. Ligation of the IMA position has two options: Not retain the left colonic artery from IMA, 1~ 2 cm above the root separation and ligation; retain the left colonic artery, separation of the left colonic artery branches in IMA distal and low ligation. We designed the clinical trials intends to carry out a large sample of a single blind, randomized controlled trial design; comparison of therapeutic effects of L-DIXON surgery of inferior mesenteric artery low ligation and high ligation, clarify the different surgical methods for patients with operation difficulty and the short-term prognosis, to investigate the best surgical method of laparoscopic radical surgery for rectal cancer. We Plan to carry out a multi-center prospective randomized controlled study, the patients were randomly divided into two groups, the control group (traditional group) and the experimental group (Retain left colic artery group). The anastomotic complications were analyzed and observed 3 years recurrence rate and survival time of postoperative patients after operation, to explore the retention of left colonic artery for guarantee can effectively reduce anastomotic complications related to the premise of oncologic under laparoscopic left colon clear retention artery in low rectal cancer surgery advantage
Joul Kassis
Damascus University, Australia
Title: Effectiveness of Chinese acupuncture on pain relief following surgical removal of impacted third molars: A self-controlled clinical trial
Time : 16:20-16:45
Biography:
Joul Kassis has completed his Bachelor degree in Dentistry from Damascus University and Master’s degree in Oral and Maxillofacial Surgery (OMFS) from Department of Oral and Maxillofacial Surgery at Damascus University. He was the Head of Doctors in Department of OMFS. He has publication in the Journal of Oral and Maxillofacial Surgery, Medicine and Pathology. Now, he is a member of the Australian Dental Association, New South Wales branch, Australia
Abstract:
Introduction: The extraction of the third molar commonly produces severe pain, swelling, and functional disability. The most common treatment for postoperative pain is non-steroidal anti-inflammatory drugs. Unfortunately, these medications are associated with systematic side effects. Acupuncture as a physical therapy may provide effective pain relief without these side effects.
Aim: This research is a self-controlled clinical trial designed to evaluate the effectiveness of Chinese acupuncture treatment in pain relief following the surgical extraction of impacted third molars.
Materials & Methods: Acupuncture treatment was applied to 50 patients for 20 min immediately fol-lowing surgical extraction of all impacted third molars on the left side of all patients. Pain assessment was performed using a standardized questionnaire that contained information about the name and age of the patient, numbering and classification of the impacted tooth, timing and duration of the surgical operation, and a table with the visual analogue scale (VAS) for measuring pain intensity. The pain intensity was measured several times. The measurement was started by the author himself who did the first two measurements. The patient was then asked to continue evaluating the pain intensity for the following periods: after 6 and 12 h and on the second, third, fourth, fifth, sixth, and seventh days.
Results: Pain intensity values were less for the acupuncture group for all studied periods.
Conclusion: Acupuncture treatment following surgical extraction of impacted third molars could reduce postoperative pain values
Anders Hoegh
Odense University Hospital, Denmark
Title: Differentiation between benign and malignant rectal tumors by dual energy CT; A pilot study
Time : 16:45-17:05
Biography:
Anders Hoegh is a qualified Surgeon. He is a member of Odense University Hospital (OUH) research unit in Surgical department. The research unit focuses on prevention, early recognition and treatment of advanced colorectal polyps and early colorectal cancer
Abstract:
Aim: Aim of this study is to assess the performance of Dual Energy Computed Tomography (DECT) in the differentiation between malignant and benign tumors in the rectum.
Methods: We enrolled eight patients with rectal tumors suspected of rectal cancer during colonoscopy. All patients underwent Computed Tomography (CT), Magnetic Resonance Imaging (MRI) and Endo-rectal Ultrasound (ERUS) for clinical staging. Furthermore, all patients underwent DECT of the pelvis. The eight patients had histopathological verified benign adenomas after transanal endoscopic microsurgery resection (TEM). The eight patients were matched with eight consecutively selected patients with histopathologically verified malignant rectal tumors. The DECT images were analyzed to assess the difference between malignant and benign rectal tumors. Experienced radiologist reviewed all DECT images. In each tumor, we applied three regions of interest (ROIs) for the acquisition of the DECT unique quantitative parameters; the mean atomic mass (effective Z value), iodine concentration, dual energy ratio (DER) and dual energy index (DEI) was determined in both groups.
Results: The comparison of all four quantitative parameters among the two groups showed a higher value of effective Z, iodine concentration and DER in malignant compared to benign tumors. In the case of the DEI, the values were lower in malignant tumors compared to benign tumors.
Conclusion: Dual energy CT may have a potential to differentiate between malignant and benign rectal tumors by using its unique and objective quantitative parameters
Madhura Joshi
Lupin Bioresearch Center, India
Title: Understanding of study endpoints for successful completion of clinical trials
Time : 17:05-17:30
Biography:
Madhura Joshi is a Medical Graduate from Bharati Vidyapeeth College and did her Master’s in Business Management in Human Resources. She also completed her Post-graduate diploma in Clinical Research and Clinical Data Management. She has more than 9 years of experience in the Clinical Research and has conducted many national/ multi-national clinical trials and designed more than 20 trial protocols. She played an instrumental role in establishing a clinical endpoint team at Lupin Bioresearch Center. Currently, she heads the Clinical Endpoint team at Lupin Bioresearch Center and designs & conducts clinical studies for the products formulated by Lupin’s generics research group and the advanced drug delivery group. She is, thus, an integral part of the Lupin team which works exclusively in identifying and understanding the clinical aspects of potential generic pharmaceutical products
Abstract:
For every clinical trial- clear hypothesis, endpoints and study design should be adequate so that study can be easily get regulatory approval. The rate of late stage clinical trial failures is the single biggest determinant of returns on pharmaceutical R&D. If those trials fail (whether for safety or lack of efficacy), all the capital invested up to that point is lost. Thus, understanding of the study endpoints is very important for study success. Study endpoints can vary as per the molecule for which the study is planned and also the therapeutic indication for which it is to be used. Clinical endpoint generally refers to occurrence of a disease, symptom, sign or abnormal laboratory finding that account for one of the target outcomes of the clinical trial. The clinical trial may have one or more clinical endpoints. Endpoints are defined as a measure that allows us to decide whether the null hypothesis of Clinical trial should be accepted or rejected. A clinical trial may have one or more (secondary) endpoints. The primary endpoint of a clinical trial is the endpoint for which subjects are randomized and are essential to establish effectiveness for approval. Secondary endpoints may be used to support the primary endpoints and for which the trial may not be powered nor randomized. As recognized there are three types of Endpoints which is direct, surrogate and composite endpoints. Direct endpoint measure how a patient feels, functions or survives. A surrogate endpoint is a measure of effect of a specific treatment that may correlate with a real clinical endpoint but does not necessarily have a guaranteed relationship. In composite endpoint, different outcomes are combined into one group. The purpose of this paper is to understand various end points used for different therapeutic indications for particular phase of trial
Elaine Walsh
University College Dublin, Ireland
Title: Factors affecting patient participation in clinical trials in Ireland: A narrative review
Time : 17:30-17:55
Biography:
Elaine Walsh completed her BSN (2008), Graduate Diploma in Adult Oncology (2012) and Master’s in Clinical Practice (2014) at University College Dublin. She has worked primarily with adult cancer patients in Ireland. She developed her passion for nursing research while working as a Clinical Research Nurse. She has published one paper (2016), and has participated in delivering webinar education to academics and healthcare professionals working in the area of clinical research
Abstract:
Objective: Clinical trials have long been considered the ‘gold standard’ of research-generated evidence in health care. Patient recruitment is an important determinant in the success of the trials, yet little focus is placed on the patient’s decision-making process towards recruitment. Our objective was to identify the key factors pertaining to patient participation in clinical trials, to better understand the identified low participation rate of patients in one clinical research facility within Ireland.
Design: Identifying studies from four databases, this narrative literature review focuses on factors, which may act to facilitate or deter patient participation in clinical trials.
Results: 61 studies were included in the narrative review: 48 of these papers focused specifically on the patient's perspective of participating in clinical trials. The remaining thirteen related to carers, family and health care professional perspectives of participation. The primary factor influencing participation in clinical trials amongst patients was related to personal factors and these were collectively associated with obtaining a form of personal gain through participation. Cancer was identified as the leading disease entity included in clinical trials.
Conclusion: The vast majority of literature relating to participation in clinical trials emanates predominantly from high-income countries, with 63% originating from the USA. No studies for inclusion in this review were identified from low income or developing countries and therefore limits the generalizability of the influencing factors


















